| ID | 220 |
| Problem No. | 2679 | Equation | .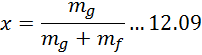 |
| Statement | (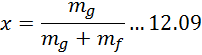 ) ) |
| Ans. (a) | (a). |
| Ans. (b) | (b). |
| Ans. (c) | (c). |
| Ans. (d) | (d). |
| Subtopic | |
| Topic | |
| Subject | Mass/Energy Balances and Thermodynamics |
| ID | 220 |
| Problem No. | 2679 | Equation | .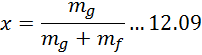 |
| Statement | (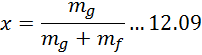 ) ) |
| Ans. (a) | (a). |
| Ans. (b) | (b). |
| Ans. (c) | (c). |
| Ans. (d) | (d). |
| Subtopic | |
| Topic | |
| Subject | Mass/Energy Balances and Thermodynamics |